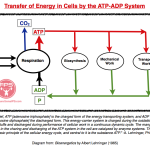
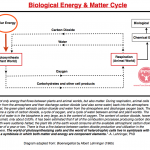
Also see:
How cancer cells rewire their metabolism to survive
Tumor Bearing Organisms – Lipolysis and Ketogenesis as Signs of Chronic Stress
Fatty Acid Synthase (FAS), Vitamin D, and Cancer
Free Fatty Acids Suppress Cellular Respiration
PUFA Promote Cancer
PUFA Decrease Cellular Energy Production
Israeli Paradox: High Omega -6 Diet Promotes Disease
Low Blood Sugar Basics
Dietary PUFA Reflected in Human Subcutaneous Fat Tissue
Towards a morphogenetic perspective on cancer
Israeli Paradox: High Omega -6 Diet Promotes Disease
Toxicity of Stored PUFA
PUFA Accumulation & Aging
The Randle Cycle
Maternal PUFA Intake Increases Breast Cancer Risk in Female Offspring
Maternal Ingestion of Tryptophan and Cancer Risk in Female Offspring
Polyamines and Cancer
Carrageenan, Inflammation, Cancer, Immunity
AIDS patients – High PUFA and High Cortisol in Blood
Cancer Cells Are What They Eat, and It’s Not Glucose
Sugar Feeds Thyroid
https://www.youtube.com/watch?v=S8o8GvyGRYc
Quotes by Ray Peat, PhD:
“Starving for sugar causes the body to break down proteins to make glucose, weakening the organism’s resistance, providing amino acids for tumor growth, and probably providing ammonia and other things that stimulate growth and interfere with differentiation. The liberated free fatty acids in either sugar deprivation or diabetes (in which cells are starved for glucose) stimulate tumor growth.”
“There are many people currently recommending fish oil (or other highly unsaturated oils) for preventing or treating cancer, and it has become almost as common to recommend a sugar free diet, “because sugar feeds cancer.” This is often, incorrectly, said to be the meaning of Warburg’s demonstration that cancer cells have a respiratory defect that causes them to produce lactic acid from glucose even in the presence of oxygen. Cancer cells use glucose and the amino acid glutamine primarily for synthetic purposes, and use fats as their energy source;the growth stimulating effect of the “essential fatty acids” (Sueyoshi and Nagao, 1962a; Holley, et al., 1974) shows that depriving a tumor of those fats retards its growth. The great energetic inefficiency of the cancer metabolism, which causes it to produce a large amount of heat and to cause systemic stress, failure of immunity, and weight loss, is because it synthesizes fat from glucose and amino acids, and then oxidizes the fat as if it were diabetic.”
“”Cancer metabolism” or stress metabolism typically involves an excess of the adaptive hormones, resulting from an imbalance of the demands made on the organism and the resources available to the organism. Excessive stimulation depletes glucose and produces lactic acid, and causes cortisol to increase, causing a shift to the consumption of fat and protein rather than glucose. Increased cortisol activates the Randle effect (the inhibition of glucose oxidation by free fatty acids), accelerates the breakdown of protein into amino acids, and activates the enzyme fatty acid synthase, which produces fatty acids from amino acids and pyruvate, to be oxidized in a “futile cycle,” producing heat, and increasing the liberation of ammonia from the amino acids. Ammonia suppresses respiratory, and stimulates glycolytic, activity.”
“In Warburg’s view, cancer has a unique requirement for glucose, because of its “respiratory defect.” The ability of cancer to consume large amounts of glucose means that the body interprets cancer as stress, since the waste of glucose is the key feature of stress. The body’s response to decreasing glucose is to produce larger amounts of adrenal hormones, especially cortisol. Cortisol raises the blood glucose supply by converting tissue proteins into sugar. This hormonal response to the sugar appetite of cancer is the main cause of the wasting syndrome which makes cancer so serious, and which destroys the thymus gland, causing the immune system to lose its guidance. V. S. Shapot’s work, showing the involvement of glucose and cortisol in cancer, provides part of the context for understanding the implications of Warburg’s view of cancer as a “respiratory defect.””
“Ketones are very protective as a fuel, but the problem is that they are produced as a result of metabolic stress. If the liver is extremely good, it can store enough glycogen for a day, but chronic, frequent, stress usually damages the liver’s ability to store glycogen.”
Cancer Res. 1987 Feb 15;47(4):1065-8.
Blood nutrient concentrations and tumor growth in vivo in rats: relationships during the onset of an acute fast.
Sauer LA, Dauchy RT.
The rate of tumor growth in vivo in adult rats (250- to 350-g total body weight) is stimulated during an acute fast. No tumor growth stimulation is observed in fasted immature rats (less than about 200-g total body weight). The different tumor growth responses in rats of these two age groups appear to depend on the increased availability to the tumor of nutrients from host fat stores in adult rats. Immature rats, which lack significant fat stores, show neither hyperlipemia nor ketosis during fasting. These experiments were performed to determine the relationship between blood fat store-derived nutrient concentrations and the onset of stimulated tumor growth in fasted adult rats. Animals were matched for tumor size and growth during a period of ad libitum feeding preceding the fast. Tumor growth was documented by increased size and incorporation of [methyl-3H]thymidine into tumor DNA. Mobilization of host fat stores leading to increased blood concentrations of free fatty acids, glycerol, ketone bodies, and triglycerides started about 7 h after food was removed and reached its maximum after about 15 h. Increased rates of tumor growth and incorporation of thymidine into tumor DNA correlated closely with the higher circulating nutrient concentrations. Both the nutrient concentrations and tumor growth were decreased by refeeding. These findings suggest that the availability of nutrients derived from host fat stores may be rate limiting for tumor growth in vivo.
Cancer Res. 1986 Jul;46(7):3469-75.
Stimulation of tumor growth in adult rats in vivo during an acute fast.
Sauer LA, Nagel WO, Dauchy RT, Miceli LA, Austin JE.
These experiments investigate an increase in tumor growth that occurs in adult rats in vivo during an acute fast. The effects of feeding, fasting, and underfeeding on the growth of Morris hepatomas 5123C and 7288CTC in Buffalo rats and of Walker carcinoma 256 and Jensen sarcoma in Sprague-Dawley rats were studied. Animals were matched for tumor size and growth during a period of ad libitum feeding preceding the fasting or underfeeding. Tumor growth was documented by increased size and incorporation of [methyl-3H]thymidine into tumor DNA. Fasting increased the rate of growth of the tumors 3 to 4 times over that measured in fed rats. This effect began during the first day of fasting and ended abruptly on refeeding. After refeeding tumor growth slowed to the rate in fed rats. Tumors from fed or fasted rats were not different in cellularity or dry weight/g wet weight. A positive growth response in the tumor required lipolysis and ketosis in the host. No stimulation was observed during an acute fast in either immature rats or in mature rats whose weights had been reduced by underfeeding. These animals have small fat stores and show no increase in arterial blood free fatty acid or ketone body concentrations during an acute fast. Finally, underfeeding of adult rats raised the blood concentrations of these nutrients to values that were intermediate between those in fasted and fed rats. Tumor growth rates in these rats were intermediate between those in fasted and fed rats. The results support the proposal that an increase in availability of free fatty acids and/or ketone bodies is the stimulus that increases the rate of tumor growth during an acute fast.
Cancer Res. 1987 Apr 1;47(7):1756-61.
Stimulation of tumor growth in adult rats in vivo during acute streptozotocin-induced diabetes.
Sauer LA, Dauchy RT
The effects of acute diabetes mellitus on the growth of Morris hepatoma 7288CTC and Jensen sarcoma were studied in fed, young (less than 200 g), and adult (greater than 250 g) rats. Animals were matched for tumor size and growth; the rates of tumor growth were the same in fed, young and adult nondiabetic rats. Diabetes was induced by the i.v. injection of streptozotocin (65 mg/kg total body weight) into tumor-bearing rats and changes in arterial blood nutrient concentrations were compared to changes in the rates of tumor growth and DNA synthesis. In young rats acute diabetes did not increase the blood concentrations of the fat store-derived nutrients and did not increase the rate of tumor growth. In adult rats, however, acute diabetes raised the arterial blood free fatty acid, glycerol, triglyceride, and ketone body concentrations to high levels and increased the rate of tumor growth about three times over that observed in untreated rats. Progress curves for the mobilization of host fat stores and for incorporation of [methyl-3H]thymidine into tumor DNA during the onset of diabetes showed that these activities were closely correlated in adult rats. Both processes began to increase 2 to 4 h after streptozotocin treatment, reached an initial peak at 12 to 16 h, decreased to a low point at 18 to 20 h, and then increased again to the new steady state after 23 to 24 h. The results indicate that the rate of tumor growth in rats in vivo is limited by the availability of a substance(s) present in the hyperlipemic blood of adult diabetic rats. The tight relationship between host lipolysis and tumor growth suggests that the substance(s) is derived from host fat stores.
Cancer Res. 1988 Jun 1;48(11):3106-11.
Identification of linoleic and arachidonic acids as the factors in hyperlipemic blood that increase [3H]thymidine incorporation in hepatoma 7288CTC perfused in situ.
Sauer LA, Dauchy RT.
Tumor growth and the incorporation of [3H]thymidine into tumor DNA in vivo are increased about 3 times in adult rats (greater than 250 g) after 1 to 2 days of starvation or the induction of diabetes with streptozotocin. These tumor growth responses require hyperlipemia and are reversed by refeeding or insulin treatment, respectively. They do not occur in young tumor-bearing rats (less than about 150 g) that lack appreciable fat stores. A direct relationship between the increased rates of both [3H]thymidine incorporation and tumor growth and host hyperlipemia suggests that tumor cell renewal in vivo in fed rats is limited by substances that are present in hyperlipemic blood. In this study we used a procedure for perfusion of solid tumors in situ to measure the sensitivity of tumor [3H]thymidine incorporation to hyperlipemic blood and to identify the rate-limiting substances. Tissue-isolated Morris hepatomas (7288CTC) growing in young or adult Buffalo rats were perfused with blood from donor rats. Hyperlipemic blood for perfusion was obtained from 2-day starved tumor-bearing (Buffalo) or non-tumor-bearing (Buffalo or Lewis) rats. At the end of the perfusions the tumors were labeled with a pulse of [3H]thymidine (2 microCi/g estimated tumor wet weight). [3H]Thymidine incorporation in tumors growing in fed adult rats was increased from 80 +/- 5 (SD) dpm/micrograms DNA at zero time (before perfusion) to 209 +/- 9 dpm/micrograms DNA (n = 3) after perfusion for 3 h. Tumors growing in fed or starved young rats showed similar responses, and hyperlipemic blood from non-tumor-bearing rats was as effective as hyperlipemic blood from tumor-bearing rats. Perfusion of tumors growing in starved rats with normolipemic blood from fed adult rats decreased [3H]thymidine incorporation from 211 +/- 13 dpm/micrograms DNA before perfusion to 68 +/- 9 dpm/micrograms DNA (n = 3) after perfusion for 3 h. Cells, plasma, and plasma subfractions from hyperlipemic blood were reconstituted to whole blood using plasma, cells, and whole blood, respectively, from fed rats and the mixtures were perfused into tumors growing in fed adult rats. Mixtures containing hyperlipemic plasma, lipid extracts (ethanol:acetone, 1:1) of hyperlipemic plasma, or albumin from hyperlipemic plasma increased tumor [3H]thymidine incorporation. Free fatty acid concentrations were increased about five times in hyperlipemic plasma and perfusion of tumors with normolipemic blood containing added linoleic and arachidonic acids increased [3H]thymidine incorporation. Blood mixtures containing palmitic, stearic, and oleic acids were inactive.(ABSTRACT TRUNCATED AT 400 WORDS)
J Nutr. 1997 Jul;127(7):1412-21.
Dietary linoleic acid intake controls the arterial blood plasma concentration and the rates of growth and linoleic acid uptake and metabolism in hepatoma 7288CTC in Buffalo rats.
Sauer LA, Dauchy RT, Blask DE.
In this study, we tested the hypothesis that dietary linoleic acid intake controls the arterial blood plasma linoleic acid concentration and the rates of tumor growth and linoleic acid metabolism in vivo. Seven groups of young male Buffalo rats (11-21 rats/group) were given free access to semipurified diets containing different amounts of corn and/or olive oils. Four other groups (7-11 rats/group) were 30% energy-restricted. Each experiment included periods for rat growth and plasma lipid stabilization (6 wk), measurement of mean daily arterial blood plasma fatty acid concentrations (3 wk), surgical implantation of a subcutaneous tissue-isolated hepatoma 7288CTC, tumor growth and harvest (2-4 wk). Linoleic + arachidonic acid (P = 0.007) and oleic acid (P = 0.002) concentrations in arterial blood plasma were increased as dietary intake of linoleic and oleic acids was increased, respectively. In rats given free access to food, tumor growth was directly dependent on the plasma concentrations of linoleic (P < 0.001) and arachidonic acids (P = 0.04). Tumor growth in energy-restricted rats was dependent only on the linoleic acid concentration (P = 0.008). Energy restriction itself caused a growth inhibition independent of plasma linoleic acid. The linoleic acid and total fatty acid concentrations of tumor triacylglycerols were directly dependent on the plasma linoleic acid concentration in rats given free access to food (P = 0.009). Hepatoma 7288CTC (both in vivo and during perfusion in situ) supported a dose-dependent conversion (P < 0.001) of plasma linoleic acid to the mitogen, 13-hydroxy-9, 11-octadecadienoic acid. We conclude that increased arterial blood plasma linoleic acid concentrations, caused by increased dietary intakes, specifically stimulate growth, lipid storage and linoleic acid metabolism in hepatoma 7288CTC in vivo.
Cancer Res. 1983 Aug;43(8):3497-503.
Ketone body, glucose, lactic acid, and amino acid utilization by tumors in vivo in fasted rats.
Sauer LA, Dauchy RT.
Arteriovenous differences for acetoacetate, beta-hydroxybutyrate, glucose, lactic acid, and glutamine and other amino acids were measured across Morris hepatomas 5123C, 7777, and 7288CTCF and Walker sarcocarcinoma 256 in vivo in rats fasted for 2 days. The acetoacetate and beta-hydroxybutyrate concentrations in arterial whole blood of fasted tumor-bearing rats were 0.52 +/- 0.06 and 1.82 +/- 0.19 mM (S.E., n = 38), respectively. Both ketone bodies were utilized by the tumors, and the rates of utilization were directly related to the rates of supply. The mean utilization rates for acetoacetate and beta-hydroxybutyrate were 13.9 +/- 2.9 (range, 0 to 64; n = 30) and 24.7 +/- 4.4 (range, 0 to 145; n = 38) nmol/min/g tumor wet weight, respectively. Eight of the tumors produced acetoacetate, presumably from utilized beta-hydroxybutyrate. An average of 52% of the acetoacetate and 30% of the beta-hydroxybutyrate carried in the arterial blood was removed during one pass through the tumors. The concentrations of glucose and glutamine in the arterial whole blood of fasted tumor-bearing rats (n = 38) were 6.55 +/- 0.3 and 0.76 +/- 0.02 mM, respectively; both of these substrates were utilized at rates that were directly proportional to the rates of supply. The mean rates of glucose and glutamine utilization for all tumors in fasted rats were 101 +/- 11 (range, 3 to 313) and 8.2 +/- 1.1 (range, 0 to 25.1) nmol/min/g tumor wet weight, respectively. Thirty-six % of the glucose and 25% of the glutamine supplied to the tumors was utilized. Comparison (by linear regression and analysis of covariance) of the rates of supply and utilization of glucose and glutamine in tumors growing in fasted versus fed rats indicated that these substrates are utilized more efficiently by tumors growing in fasted animals. Lactic acid was either produced or utilized, depending on the arterial whole-blood concentration. Production or utilization occurred, respectively, when the arterial lactate concentration was less or greater than 1 to 3 mM. The arterial whole-blood amino acids (except glutamine) were utilized at rates that ranged from 1 to 4 nmol/min/g tumor wet weight. The results indicate that energy production for tumor growth in fasted rats is supported, in part, by an increased availability of ketone bodies, by an increased efficiency of utilization of glucose and glutamine, and, under certain circumstances, by utilization of lactic acid.
Cell Cycle. 2011 Apr 15;10(8):1271-86. doi: 10.4161/cc.10.8.15330.
Ketones and lactate increase cancer cell “stemness,” driving recurrence, metastasis and poor clinical outcome in breast cancer: achieving personalized medicine via Metabolo-Genomics.
Martinez-Outschoorn UE, Prisco M, Ertel A, Tsirigos A, Lin Z, Pavlides S, Wang C, Flomenberg N, Knudsen ES, Howell A, Pestell RG, Sotgia F, Lisanti MP.
Previously, we showed that high-energy metabolites (lactate and ketones) “fuel” tumor growth and experimental metastasis in an in vivo xenograft model, most likely by driving oxidative mitochondrial metabolism in breast cancer cells. To mechanistically understand how these metabolites affect tumor cell behavior, here we used genome-wide transcriptional profiling. Briefly, human breast cancer cells (MCF7) were cultured with lactate or ketones, and then subjected to transcriptional analysis (exon-array). Interestingly, our results show that treatment with these high-energy metabolites increases the transcriptional expression of gene profiles normally associated with “stemness,” including genes upregulated in embryonic stem (ES) cells. Similarly, we observe that lactate and ketones promote the growth of bonafide ES cells, providing functional validation. The lactate- and ketone-induced “gene signatures” were able to predict poor clinical outcome (including recurrence and metastasis) in a cohort of human breast cancer patients. Taken together, our results are consistent with the idea that lactate and ketone utilization in cancer cells promotes the “cancer stem cell” phenotype, resulting in significant decreases in patient survival. One possible mechanism by which these high-energy metabolites might induce stemness is by increasing the pool of Acetyl-CoA, leading to increased histone acetylation, and elevated gene expression. Thus, our results mechanistically imply that clinical outcome in breast cancer could simply be determined by epigenetics and energy metabolism, rather than by the accumulation of specific “classical” gene mutations. We also suggest that high-risk cancer patients (identified by the lactate/ketone gene signatures) could be treated with new therapeutics that target oxidative mitochondrial metabolism, such as the anti-oxidant and “mitochondrial poison” metformin. Finally, we propose that this new approach to personalized cancer medicine be termed “Metabolo-Genomics,” which incorporates features of both 1) cell metabolism and 2) gene transcriptional profiling. Importantly, this powerful new approach directly links cancer cell metabolism with clinical outcome, and new therapeutic strategies for inhibiting the TCA cycle and mitochondrial oxidative phosphorylation in cancer cells.
Cancer. 1988 Feb 15;61(4):689-701.
Peripheral blood lymphocytes from patients with cancer lack interleukin-2 receptors.
Hakim AA.
When tumor cells develop in healthy adults, they activate the cellular immune system–natural killer (NK) cells, antigen-specific cytotoxic lymphocytes (CTL), and the synthesis of antigen specific cytotoxic antibodies. These are aimed at killing the intruding cells. However, in cancer patients the tumor continues to grow. As tumor cells proliferate, they were shown to release factors that mediate the inactivation of the host immune defense systems. The study documented in this article examined peripheral blood lymphocytes, mononuclear cells (MNC), NK cells, T-helper cells (THC). This study confirmed the interaction of the released inhibitor factors with these mononuclear cells. NULL cells from healthy adults responding to interleukin-2 (IL-2) and NILL cells from patients with metastatic breast carcinoma nonresponsive to IL-2 were also isolated by the standard antibodies-pinning technique. The cells were obtained from age-matched subjects: ten healthy adults; ten patients each from Stage I, II, III, and IV metastatic breast carcinoma (BCa-I, BCa-II, BCa-III, and BCa-IV or MBCa); and ten patients with benign breast disease (BBD). The responsiveness of these THC, PBMNC, NK, NULL, and NILL cells in vitro to graded levels of phytohemagglutinin (PHA), Concanavalin A (Con A), and recombinant interleukin-2 (rIL-2) was examined. Responsiveness was monitored by 3H-thymidine (3H-TdR) uptake, production and release of IL-2, interleukin-2 receptor (IL-2R), and cytotoxic activities against K-562 cells and breast carcinoma short-term cell lines. A lack of functional IL-2R in peripheral blood lymphocytes from patients with metastatic breast carcinoma was confirmed by nonsignificant anti-Tac antibody binding. An elevation in the expression of cell surface antigen GP-120 has been observed to be associated with the activation in vitro of T-cells from healthy adults and from patients with benign breast disease, but not of T-cells from patients with breast carcinoma. Biochemical studies of the GP-120 using high performance liquid chromatography combined with nitrocellulose blotting confirmed that the glycoprotein was resistant to trypsin and chymotrypsin, but susceptible to pronase. It contained sialic acid and lactosaminoglycan as O-linked sugars. It could be labeled with pariodate/NaB(3H4) and is recognized by MAbT-305 monoclonal antibodies. It contained sialic acid linked (2—3) to galactose.(ABSTRACT TRUNCATED AT 400 WORDS)
When cancer cells are implanted into a healthy host, they seldom grow, but when implanted into a host who already has cancer, they grow. (Hakim, 1988.) The healthy host provides many restorative factors, the sick host provides additional harmful factors, but few restorative factors (Chekulaev, et aI., 1987.) -Ray Peat, PhD
Biokhimiia. 1987 Sep;52(9):1501-11.
[Activation of lipolysis and ketogenesis in tumor-bearing animals as a reflection of chronic stress states].
[Article in Russian]
Chekulaev VA, Shelepov VP, Pasha-zade GR, Shapot VS.
In order to elucidate the peculiarities of brain metabolism in tumour-bearing organisms, the arterio-venous (A-V) content of glucose, acetoacetate (Ac-Ac), beta-hydroxybutyrate (beta-HB) and non-esterified fatty acids (NEFA) in growing Zajdela ascite hepatoma (ZAH) and solid hepatoma 27 (H-27) was compared. Analysis of metabolic patterns of healthy, starving and fed recipients (ZAH and H-27) revealed the inadequacy of the concepts on anorexia as being the cause of carbohydrate-lipid metabolic disturbances. In tumour-bearing organisms lipolysis and ketogenesis reflect the tumour-induced chronic stress. Absorption of beta-HB and release of Ac-Ac by brain were observed at all stages of malignant growth. This is probably due to a partial switch-over of brain metabolism to non-carbohydrate energy sources. Besides, certain stages of tumour growth are associated with active assimilation of NEFA by brain. A correlation between the A-V difference with respect to glucose and Ac-Ac as well as between the glucose and NEFA contents was established. It was assumed that the A-V difference in glucose is the main regulator of ketone body metabolism.
Prog Clin Biol Res. 1983;111:89-109.
Energy metabolism in trauma and sepsis: the role of fat.
Wolfe RR, Shaw JH, Durkot MJ.
There seems little doubt that there are signals for the increased mobilization of fat in shock, trauma, and sepsis. Whether those signals are reflected by an actual increase in mobilization is dependent on many variables including cardiovascular status. A hypothetical scheme based on our own experiments in the hyperdynamics phases of response to burn injury and to sepsis is presented in Figure 8. According to this scheme, catecholamines stimulate lipolysis in the adipose tissue, resulting in the release of glycerol and FFA into the plasma at increased rates. The glycerol is cleared by the liver and converted into glucose–a process stimulated by, among other things, glucagon. Some of the increased flux of FFA is also cleared by the liver, whereupon the fatty acids are incorporated into VLDL and released again into the plasma. The increased FFA levels also exert a dampening effect on the factors stimulating hepatic glucose production. At the periphery, plasma FFA as well as VLDL fatty acids are taken up at an increased rate. The tissues are attuned to the oxidation of fat, and as a consequence most of the energy production is derived from fat oxidation. The increased fatty acids exert an inhibitory effect on the complete oxidation of glucose, so although glucose may be taken up at an accelerated rate, the relative contribution of glucose oxidation to total energy production may fall. Rather than being completely oxidized, pyruvate is reduced to lactate and released into the plasma at an accelerated rate. The lactate then contributes to the production of glucose in the liver, completing a cyclical process called the Cori Cycle. Although all aspects of this scheme are supported by data highlighted in this paper, it certainly must be an oversimplification of the overall response of substrate metabolism to trauma and sepsis. It is presented for the purpose of highlighting the potential role of fat as a controller of the metabolic response, and to suggest that the enhanced mobilization and oxidation of fat is one of the fundamental responses to stress.
Cell Cycle. 2010 Sep 1;9(17):3506-14. Epub 2010 Sep 21.
Ketones and lactate “fuel” tumor growth and metastasis: Evidence that epithelial cancer cells use oxidative mitochondrial metabolism.
Bonuccelli G1, Tsirigos A, Whitaker-Menezes D, Pavlides S, Pestell RG, Chiavarina B, Frank PG, Flomenberg N, Howell A, Martinez-Outschoorn UE, Sotgia F, Lisanti MP.
Previously, we proposed a new model for understanding the “Warburg effect” in tumor metabolism. In this scheme, cancer-associated fibroblasts undergo aerobic glycolysis and the resulting energy-rich metabolites are then transferred to epithelial cancer cells, where they enter the TCA cycle, resulting in high ATP production via oxidative phosphorylation. We have termed this new paradigm “The Reverse Warburg Effect.” Here, we directly evaluate whether the end-products of aerobic glycolysis (3-hydroxy-butyrate and L-lactate) can stimulate tumor growth and metastasis, using MDA-MB-231 breast cancer xenografts as a model system. More specifically, we show that administration of 3-hydroxy-butyrate (a ketone body) increases tumor growth by ∼2.5-fold, without any measurable increases in tumor vascularization/angiogenesis. Both 3-hydroxy-butyrate and L-lactate functioned as chemo-attractants, stimulating the migration of epithelial cancer cells. Although L-lactate did not increase primary tumor growth, it stimulated the formation of lung metastases by ∼10-fold. Thus, we conclude that ketones and lactate fuel tumor growth and metastasis, providing functional evidence to support the “Reverse Warburg Effect”. Moreover, we discuss the possibility that it may be unwise to use lactate-containing i.v. solutions (such as Lactated Ringer’s or Hartmann’s solution) in cancer patients, given the dramatic metastasis-promoting properties of L-lactate. Also, we provide evidence for the up-regulation of oxidative mitochondrial metabolism and the TCA cycle in human breast cancer cells in vivo, via an informatics analysis of the existing raw transcriptional profiles of epithelial breast cancer cells and adjacent stromal cells. Lastly, our findings may explain why diabetic patients have an increased incidence of cancer, due to increased ketone production, and a tendency towards autophagy/mitophagy in their adipose tissue.
Brain Dev. 2010 Apr;32(4):318-22. doi: 10.1016/j.braindev.2009.04.009. Epub 2009 May 13.
Tumor growth in patients with tuberous sclerosis complex on the ketogenic diet.
Chu-Shore CJ1, Thiele EA.
PURPOSE:
New evidence is emerging that the availability of nutrients plays a key role in regulating the mammalian target of rapamycin complex-1 (mTORC1) signaling pathway in human cancers. Tuberous sclerosis complex (TSC) is a genetic disorder which results in the growth of hamartomatous lesions in multiple organs due to insufficient suppression of the mTORC1 pathway. A minority of patients with TSC who develop epilepsy which is intractable to standard anticonvulsant medical and/or surgical treatments are treated with the ketogenic diet. To provide insight into the effects of nutrient manipulation on tumor growth in this condition, we describe our experience in a unique group of patients with known tuberous sclerosis complex who are on the ketogenic diet for seizure control.
METHODS:
A retrospective chart review was performed of patients with TSC treated with the ketogenic diet between January 2002 and May 2007 at Massachusetts General Hospital.
RESULTS:
Five patients with definite TSC underwent serial imaging for tumor growth while on the ketogenic diet or had unchanged imaging prior to the onset of the diet and after termination. Three out of five patients, all children, had progression of a known tumor or tumors or the development of a new tumor while on the ketogenic diet.
CONCLUSION:
In this limited case series of five TSC patients, the ketogenic diet did not induce tumor regression or suppress the growth of TSC-related tumors.
Cell Cycle. 2012 Nov 1;11(21):3964-71. doi: 10.4161/cc.22137. Epub 2012 Sep 19.
Ketone body utilization drives tumor growth and metastasis.
Martinez-Outschoorn UE, Lin Z, Whitaker-Menezes D, Howell A, Sotgia F, Lisanti MP.
We have previously proposed that catabolic fibroblasts generate mitochondrial fuels (such as ketone bodies) to promote the anabolic growth of human cancer cells and their metastasic dissemination. We have termed this new paradigm “two-compartment tumor metabolism.” Here, we further tested this hypothesis by using a genetic approach. For this purpose, we generated hTERT-immortalized fibroblasts overexpressing the rate-limiting enzymes that promote ketone body production, namely BDH1 and HMGCS2. Similarly, we generated MDA-MB-231 human breast cancer cells overexpressing the key enzyme(s) that allow ketone body re-utilization, OXCT1/2 and ACAT1/2. Interestingly, our results directly show that ketogenic fibroblasts are catabolic and undergo autophagy, with a loss of caveolin-1 (Cav-1) protein expression. Moreover, ketogenic fibroblasts increase the mitochondrial mass and growth of adjacent breast cancer cells. However, most importantly, ketogenic fibroblasts also effectively promote tumor growth, without a significant increase in tumor angiogenesis. Finally, MDA-MB-231 cells overexpressing the enzyme(s) required for ketone re-utilization show dramatic increases in tumor growth and metastatic capacity. Our data provide the necessary genetic evidence that ketone body production and re-utilization drive tumor progression and metastasis. As such, ketone inhibitors should be designed as novel therapeutics to effectively treat advanced cancer patients, with tumor recurrence and metastatic disease. In summary, ketone bodies behave as onco-metabolites, and we directly show that the enzymes HMGCS2, ACAT1/2 and OXCT1/2 are bona fide metabolic oncogenes.
Cell Cycle. 2012 Nov 1; 11(21): 3956–3963.
Ketone bodies and two-compartment tumor metabolism: Stromal ketone production fuels mitochondrial biogenesis in epithelial cancer cells
Ubaldo E. Martinez-Outschoorn, Zhao Lin, Diana Whitaker-Menezes, Anthony Howell, Michael P. Lisanti, and Federica Sotgia
We have previously suggested that ketone body metabolism is critical for tumor progression and metastasis. Here, using a co-culture system employing human breast cancer cells (MCF7) and hTERT-immortalized fibroblasts, we provide new evidence to directly support this hypothesis. More specifically, we show that the enzymes required for ketone body production are highly upregulated within cancer-associated fibroblasts. This appears to be mechanistically controlled by the stromal expression of caveolin-1 (Cav-1) and/or serum starvation. In addition, treatment with ketone bodies (such as 3-hydroxy-butyrate, and/or butanediol) is sufficient to drive mitochondrial biogenesis in human breast cancer cells. This observation was also validated by unbiased proteomic analysis. Interestingly, an MCT1 inhibitor was sufficient to block the onset of mitochondrial biogenesis in human breast cancer cells, suggesting a possible avenue for anticancer therapy. Finally, using human breast cancer tumor samples, we directly confirmed that the enzymes associated with ketone body production (HMGCS2, HMGCL and BDH1) were preferentially expressed in the tumor stroma. Conversely, enzymes associated with ketone re-utilization (ACAT1) and mitochondrial biogenesis (HSP60) were selectively associated with the epithelial tumor cell compartment. Our current findings are consistent with the “two-compartment tumor metabolism” model. Furthermore, they suggest that we should target ketone body metabolism as a new area for drug discovery, for the prevention and treatment of human cancers.
Front. Endocrinol., 02 February 2016
Fenofibrate Induces Ketone Body Production in Melanoma and Glioblastoma Cells
Maja M. Grabacka, Anna Wilk, Anna Antonczyk, Paula Banks, Emilia Walczyk-Tytko, Matthew Dean, Malgorzata Pierzchalska, and Krzysztof Reiss
“The capacity of cancer cells to produce ketone bodies described here is quite different from previous reports, including series of papers (58–60) by the Lisanti group and coworkers regarding the possibility of ketogenesis in stromal fibroblasts surrounding breast cancer lesions. In those papers, the authors showed that cancer cells actively contributed to the stromal fibroblasts’ metabolic reprograming and took advantage of the subsequent ketone body consumption for energy generation. This is a special property of epithelia-derived tumors, such as breast carcinomas, since other cancer types do not express the proper enzymatic machinery for ketone body utilization as a source of energy (25, 28, 34). More often, cancers such as neuroblastomas or astrocytomas do absorb ketone bodies, but they use them for lipid synthesis rather than energy production (61–63).”
“In conclusion, it is quite surprising that fenofibrate is able to reprogram melanoma and glioblastoma metabolic pathways in such a way that they suffer from an energy deficit but are still forced to produce ketone bodies. This finding is important because, as far as we are aware, ketogenesis in cancer cells has not been described before and the underlying mechanism still waits to be revealed. The ketone bodies released by glioblastoma cells could serve not only as a fuel but also as a cytoprotective signaling molecule for neurons in the microenvironment surrounding the tumor tissue.”